This breakthrough could help to eradicate Ebola
In 2014, the destructive Ebola virus killed tens of thousands in west Africa and sent the world into delirium about its potential spread.
But less than two years after the deadly outbreak began, scientists have made an incredible breakthrough that could stop the virus in its tracks and confine it to the annals of history.
A new vaccine being trialled in Guinea has so far proved 100 per cent effective in clinical trials, giving hope to those tasked with stopping the virus’ spread.
• Australia’s Ebola response: we spent how much?
• Everything you need to know about Ebola
• Worse than war: my year fighting Ebola in Africa

A Red Cross worker in Sierra Leone carries a child away. The child died from Ebola. Photo: AAP
“It’s obviously promising,” the woman tasked with heading up Australia’s Ebola response – University of Sydney Professor Lyn Gilbert – told The New Daily.
“It’s almost certain to produce some measurable degree of protection,” she said.
“This may be it, because its the first time there has been confirmation of protective effect in humans.
“It’s an important breakthrough, there is no doubt about that.”
Trials of the Canadian-developed drug VSV-EBOV, were conducted in Guinea’s capital of Conakry between April and July this year.
The results of the study were released in medical journal The Lancet on Saturday.
“The premise is that by vaccinating all people who have come into contact with an infected person you create a protective ‘ring’ and stop the virus from spreading further,” Norwegian Institute of Public Health Division of Infectious Disease Control director John-Arne Røttingen said.
“This strategy has helped us to follow the dispersed epidemic in Guinea, and will provide a way to continue this as a public health intervention in trial mode.”
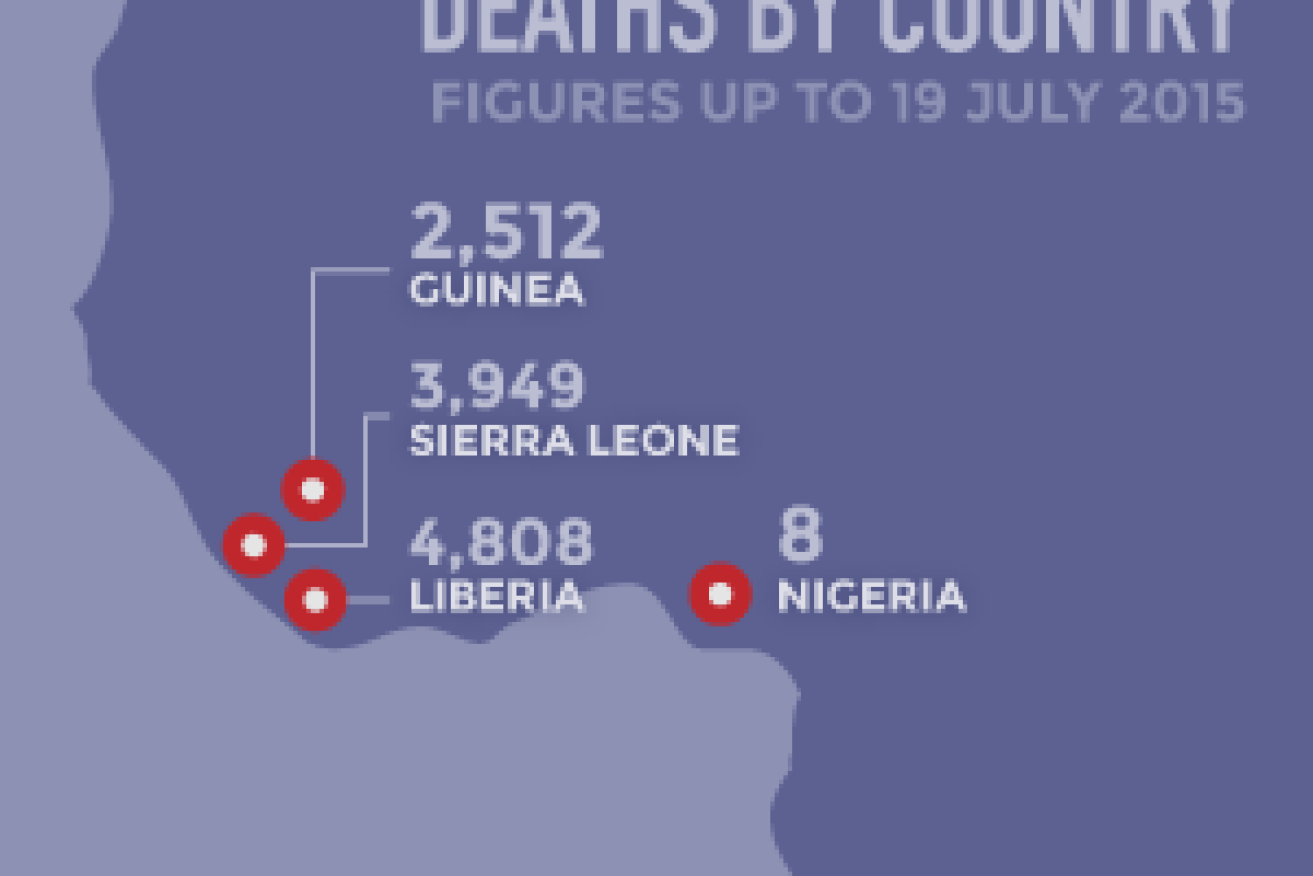
As of July 2015, the recent Ebola outbreak had killed 11,284 people and infected a total of 27,741, with the highest number of cases in Liberia, Sierra Leone and Guinea.
After spreading at an alarming rate through the latter part of 2014, the most recent outbreak began to significantly slow from March 2015.
And between June to July 2015, there was only one death from Ebola.
Still no silver bullet
Professor Gilbert was reluctant to overstate the significance of the vaccine, despite the promise of the findings.
“It is not going to solve the problem of Ebola. The problem of Ebola needs to be solved with infection control more than anything else,” she said.

Workers in full anti infectious attire lower a deceased person that died from Ebola. Photo: AAP
“But certainly its a way of protecting contacts and probably health care workers it should be at least useful.”
She also explained that it is vital more evidence is developed on a bigger sample size and over a longer period of time.
“The only reservation is the number of patients and the long-term protective effect, and I think that’s what we don’t know,” Professor Gilbert said.
“What we don’t know is what the percentage protectiveness will be and for how long. It is very difficult to predict that.”
The vaccine will be used to treat patients, but also to try and protect those coming into contact with Ebola patients.
She said it is not yet a “vaccine that’s going to be given to whole populations.”
More conclusive evidence is necessary to better understand the drug’s effectiveness.
Randomised trials have now ended, with all people at risk now able to receive the vaccine.
The study was an international collaboration between the Government of Guinea, the trial’s sponsor the World Health Organisation, Médecins Sans Frontières (Doctors Without Borders) and the Norwegian Institute of Public Health.
NewLink Genetics and American pharmaceutical company Merck currently hold the licence for the vaccine.










