French firm Servier Laboratories fined millions for deadly diet pill
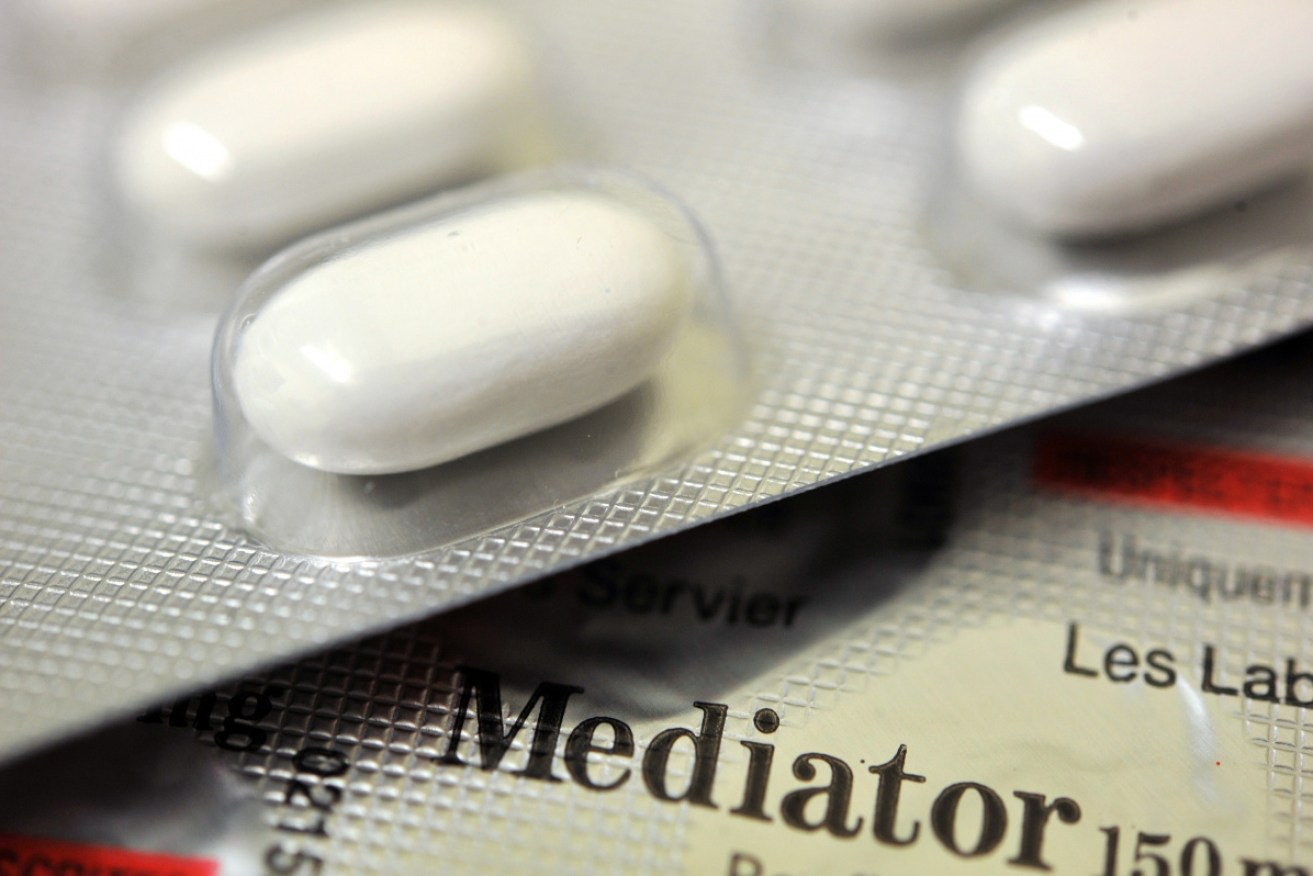
Mediator is believed to have killed 500 people over three decades in France before it was banned in 2010. Photo: Getty
A Paris court has handed millions of euros in fines to a French pharmaceutical company for its role in one of the nation’s biggest modern health scandals.
The ruling capped a judicial marathon targeting Servier Laboratories and involving more than 6500 plaintiffs, finding it guilty of manslaughter and other charges for selling a diabetes drug blamed for hundreds of deaths.
The huge trial was spread over 10 months in 2019 and 2020, and nearly 400 lawyers worked on the case.
It centred on the diabetes drug Mediator.
Servier was accused of putting profits ahead of patients’ welfare by allowing the drug to be widely and irresponsibly prescribed as a diet pill, with deadly consequences.

French pulmonologist Irene Frachon with a photo book depicting portraits of ‘‘Mediator victims’’ as she arrives at a Paris court in September 2019. Photo: Getty
Servier argued that it did not know about the drug’s dangers.
The court found Servier guilty of manslaughter, involuntary wounding and aggravated deception. The firm was acquitted of fraud.
Judges handed Servier a fine of 2.7 million euros ($4.2 million).
They were also expected to rule on plaintiffs’ demands for a total of 1 billion euros ($1.5 billion) in damages.
In the 33 years Mediator was on the market it was suspected in up to 2000 deaths among millions who took it as an appetite suppressant, according to a 2010 study.
Doctors also linked it to heart and lung problems.
One doctor flagged concerns as far back as 1998, and testified that he was bullied into retracting them.
Facing questions about the drug’s side effects from medical authorities in Switzerland, Spain and Italy, Servier withdrew it from those markets between 1997 and 2004.
Lawyers for Servier argued the company was not aware of the risks associated with Mediator before 2009, and said the company never claimed it was a diet pill. They had argued for acquittal.
It took an independent investigation by another worried French doctor before the company suspended sales in its main market in France in 2009.
The company’s CEO and founder, Jacques Servier, was indicted early in the legal process but died in 2014.
Want to see more stories from The New Daily in your Google search results?
- Click here to set The New Daily as a preferred source.
- Tick the box next to "The New Daily". That's it.








