Coronavirus tests: Portable unit gives a yes-or-no reading in five minutes

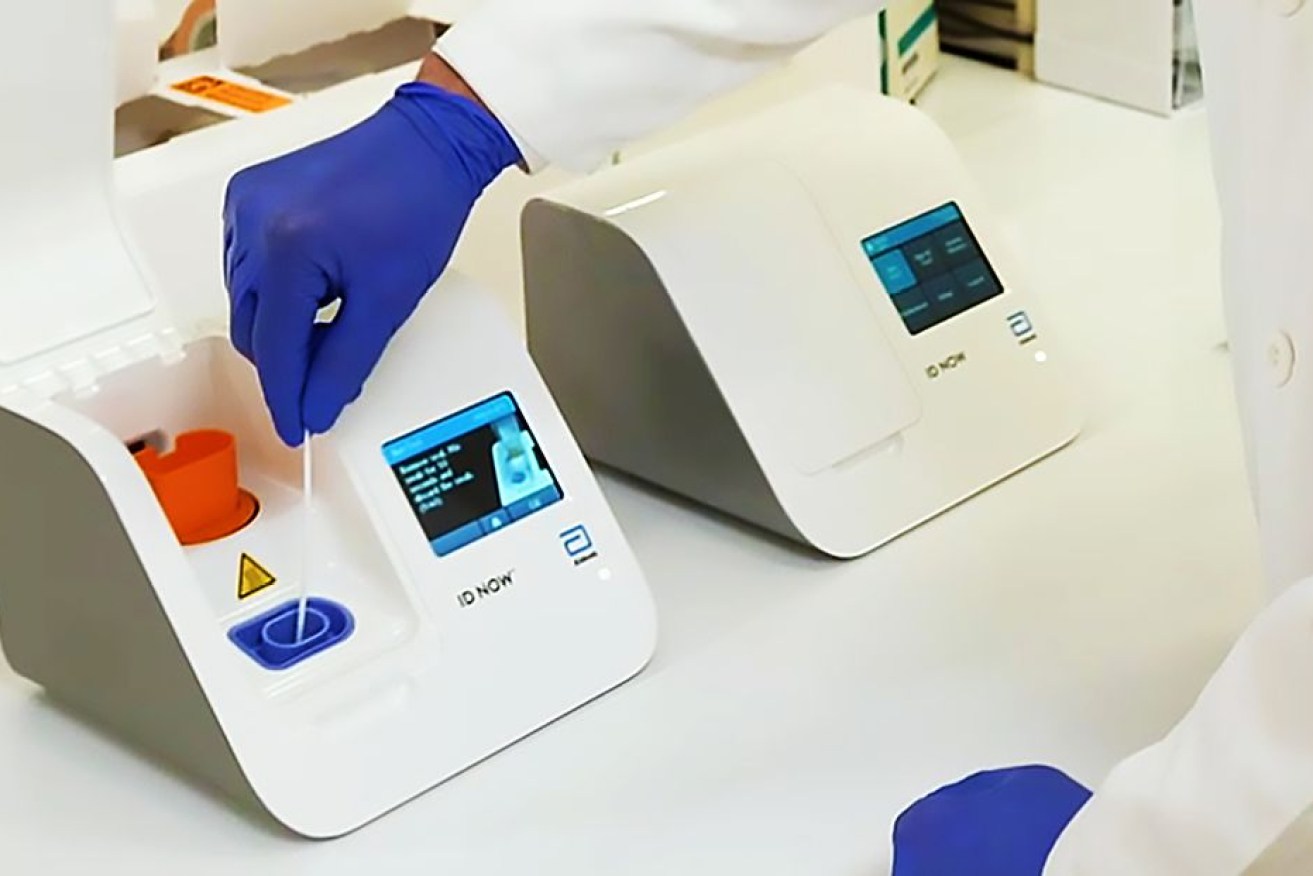
The new test for coronavirus that can be done anywhere, giving a positive result in five minutes. Photo: Abbott Laboratories
A US company has developed a point-of-care coronavirus test that can purportedly tell if someone is infected within five minutes. The device is said to be no bigger than a toaster and is thus highly portable and can be used in just about any setting.
The US Federal Drug Administration has approved the use of the test under its Emergency Use Authorisation (EUA), reflecting a trend for fast-tracking devices and drugs in the US under emergency guidelines.
According to a statement from the Abbott Laboratories, the molecular test “looks for fragments of the coronavirus genome, which can be detected in as little as five minutes when it’s present at high levels.”
“A thorough search to definitively rule out an infection can take up to 13 minutes,” the company said.
Good news! New quick test for COVID-19 approved.
15-minute test. 500,000 kits are due to land in Australia this week if all goes well with airfreight space, planes are able to land and they pass border control. A priority! #COVID19Aus— MJA Editor in Chief (@MJA_Editor) March 22, 2020
Abbot Laboratories, based in Illinois, plans to supply 50,000 tests a day from April 1. It eventually expects to produce 5 million tests a month across two systems.
- Related: PM promises $1.1 billion health package for Australians stuck at home
- Related: Trump orders General Motors to make ventilators
If the company’s claims are true – especially the ability to provide a definitive result in less than 15 minutes – the new test outperforms anything available in Australia.
Four days ago, the Therapeutic Goods Administration gave an “expedited” thumbs up to a Chinese-developed point-of-care test that is able to deliver a positive result in about 15 minutes.
Marketed by CTK Biotech, the OnSite COVID-19 IgG/IgM Rapid Test works by detecting anti-SARS-CoV-2 IgG and IgM antibodies in either human serum, plasma or whole blood “within just 10 minutes”, according to the company’s website.
“This test is easy-to-use, requires only minimally skilled personnel with basic equipment needs,” the company said.
“The OnSite COVID-19 IgG/IgM Rapid Test has 96.9 per cent sensitivity and 99.4 per cent specificity making it one of the most effective screening tests for COVID-19.”
- Related: Four treatment trials underway
A study in the peer-reviewed Journal of Medical Virology said the test could return results within 15 minutes.
The test isn’t diagnostic, but used for initial screening only. However, they don’t require laboratory processing before a result is returned. Until now, results have taken at least 24 hours.
According to a statement from the Department of Health, the approval was grant “based on evidence provided to the TGA at the time of application” – which means the TGA hasn’t assessed the test first-hand.
“All approvals are subject to conditions that additional evidence to support the ongoing safety and performance of the devices be provided to the TGA within 12 months of approval,” a department spokesman told The Guardian.
According to the TGA website, there are 16 tests for COVID-19 listed for “legal supply” in Australia. Half of them are point-of-care. Most of these approvals were given in the last week.
Ten days ago, Australia’s chief medical officer, Brendan Murphy, said the country faced “extreme pressure” supplying enough testing kits as the rate of infection continues to rise.
The federal health department at that time said there was “merit” in the COVID-19 IgG/IgM Rapid Test Kit, but local importers complained that TGA approval was proving too costly and taking too to long to be granted.
All that seems to have suddenly changed.
Will the new five-minute test, and its high degree of accuracy, be heading to Australia any time soon? No doubt importers and health officials will be keen to learn more. The main obstacle might not be TGA approval, but the fact that the US is now the leading hotspot for infections.
With no public health system to handle a runaway outbreak, and with the virus now moving inland from the east and west coasts, our good friends are in need of every tool at their disposal.








